Exactech Inc. has announced a recall of its artificial knee product because of the possibility of a degrading plastic component, an issue similar to the one that forced a recall of its artificial hips in 2021.
The company reports defective packaging may allow oxygen and other environmental contamination to degrade a key plastic component prior to implantation. Resulting oxidation can cause the plastic to prematurely wear or become damaged after implantation into the patient’s body.
The recall involves both partial and total knee replacement products, impacting more than 133,000 artificial knees manufactured and/or implanted in the United States between 2004 and 2022.
Recalled products include:
OPTETRAK® Unicondylar Tibial Components
OPTETRAK® All-polyethylene CR Tibial Components
OPTETRAK® All-polyethylene PS Tibial Components
OPTETRAK® B-Series Ps Tibial Insert
OPTETRAK® CC Tibial Inserts
OPTETRAK® CR Tibial Inserts
OPTETRAK® CR Slope Tibial Inserts
OPTETRAK® PS Tibial Inserts
CUSTOM OPTETRAK® Angled PS Insert
CUSTOM OPTETRAK® CC Tibial Insert
OPTETRAK® HI-FLEX® PS Tibial Inserts
OPTETRAK “MOMB” Non-Mod Molded Insert
OPTETRAK® RBK PS Tibial Components
OPTETRAK Logic®
OPTETRAK Logic® CR Tibial Inserts
OPTETRAK Logic® CR Slope Tibial Inserts
OPTETRAK Logic® CRC Tibial Inserts
OPTETRAK Logic® PS Tibial Inserts
OPTETRAK Logic® PSC Tibial Inserts
OPTETRAK Logic® CC Tibial Inserts
TRULIANT® CR Tibial Inserts
TRULIANT® CR Slope Tibial Inserts
TRULIANT® CRC Tibial Inserts
TRULIANT® PS Tibial Inserts
TRULIANT® PSC Tibial Inserts
Defective Knee Implant Claims in Colorado
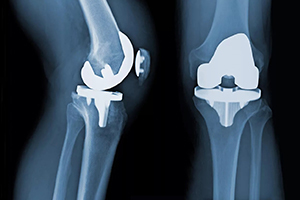
A plastic insert that fits between the femoral component and tibial component and acts as the cushion or cartilage for your replaced knee joint, known as the tibial polyethylene, is a critical component of your new knee.
A standard knee replacement has four parts:
- A metal piece that attaches to your thigh bone, known as the femoral component.
- A metal piece that fits into your shin bone, known as the tibial tray.
- A piece of plastic that fits onto your kneecap, known as the patellar component.
- Plastic that fits between the femoral component and tibial component and acts as the new cushion or cartilage for your replaced knee joint, called the tibial polyethylene insert.
The plastic inserts were packaged in vacuum bags that did not contain an additional oxygen barrier layer of Ethylene Vinyl Alcohol (EVOH). That can allow increased oxidation of the plastic, leading to a reduction of mechanical properties of the knee joint.
Premature wear or damage to the plastic component can result in pain, osteolysis bone loss, loss of mobility and the need for revision surgery or other medical procedures. While the company has established a claims process and hired a third-party administration, the process clearly leaves it in charge of determining the needs of the patient and the amount of reimbursement.
“In those cases where the plastic has worn out earlier than expected or has been damaged, we will evaluate your knee replacement and decide whether additional treatment is needed,” Exactech stated. “After this evaluation is complete, we will decide if additional treatment, including revision surgery, is necessary.”
Those dealing with defective knees or other faulty medical devices should not rely upon the company to make things right. At best, you will be reimbursed for a portion of medical expenses, without regard to pain and suffering, lost wages, reduced quality of life, or the long-term effects of patching up a defective artificial knee. Contacting an experienced defective knee implant attorney in Colorado will best help you determine your options, while protecting your rights to recover all of the compensation to which you are entitled. If you are suffering from surgical complications or issues related to a defective knee implant, please call the McLaughlin Law Firm at (720) 420-9800 for a free and confidential consultation to discuss your rights.


